The classification hierarchy differs between CLP and DPD:
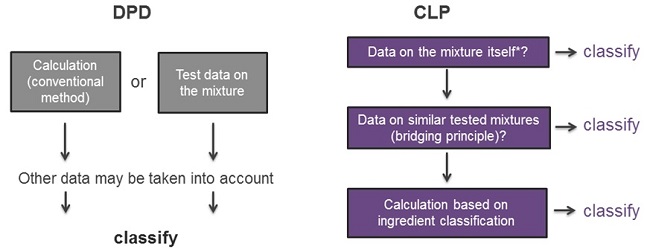
The key changes are:
- The new hierarchy of CLP (see ECHA guidelines) is clearer: for skin and eye effects, mixture data prevail. It is important to note that CLP does not allow testing on humans for classification purposes and new tests on animals can only be undertaken in certain circumstances. Existing in vivo data (animal, human) and in vitro data can be used together with any new data generated using in vitro test methods;
- Test data are not restricted to methods in Test Methods Regulation (EC) No. 440/2008: methods validated according to international procedures or based on sound scientific principles that are internationally recognised can also be used;
- Bridging principles are clearly defined in CLP Annex I. They can be regarded as rules for ‘reading across’ from one mixture to another, based on the composition of the tested and the untested mixture(s);
- Possibility to create an industry network for classification.